 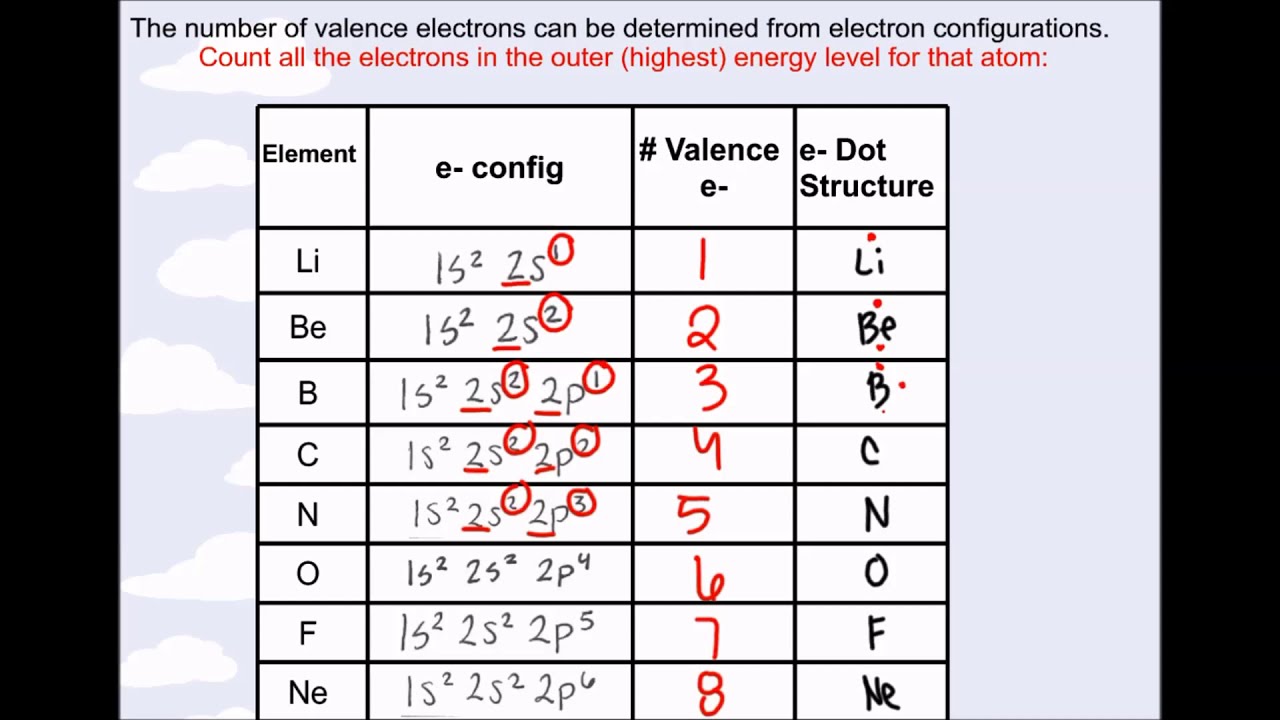
The distance between the radii is 266 p m. Beneath the molecule is the label, “B r radius equals 228 p m divided by 2 equals 114 pm.” The fourth diatomic molecule is in purple. The distance between the radii is 228 p m. These dots are arranged to the right and left and above and below the symbol, with no more than two. The number of dots equals the number of valence electrons in the atom. Beneath the molecule is the label, “C l radius equals 198 p m divided by 2 equals 99 pm.” The third diatomic molecule is in red. A Lewis electron-dot symbol (or electron-dot symbol or a Lewis symbol) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. 
The distance between the radii is 198 p m. The second diatomic molecule is in a darker shade of green. Beneath the molecule is the label, “F radius equals 128 p m divided by 2 equals 64 p m.” The next three models are similarly used to show the atomic radii of additional atoms. The distance between the centers of the two atoms is indicated above the diagram with a double headed arrow labeled, “128 p m.” The endpoints of this arrow connect to line segments that extend to the atomic radii below. Two spheres are pushed very tightly together. This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. 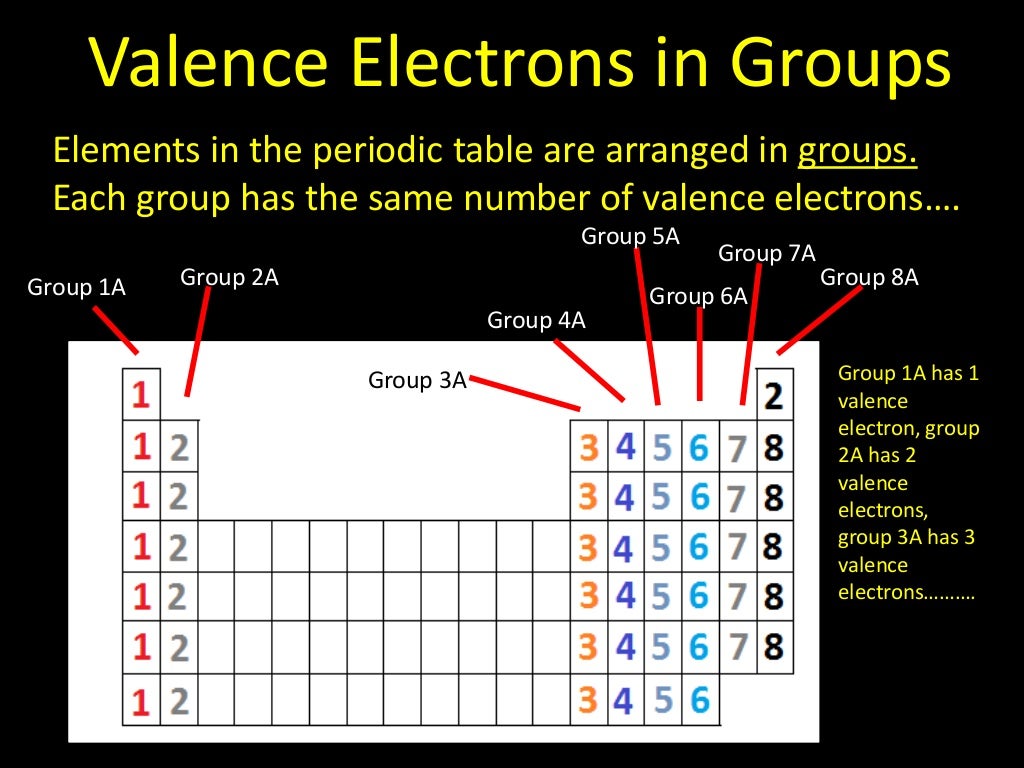
The first model, in light green, is used to find the F atom radius. However, one might think that since the number. Electron affinity decreases down the groups and from right to left across the periods on the periodic table because the electrons are placed in a higher energy level far from the nucleus, thus a decrease from its pull. In figure a, 4 diatomic molecules are shown to illustrate the method of determining the atomic radius of an atom. The less valence electrons an atom has, the least likely it will gain electrons. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. Do you notice any patterns in the number of valence electrons The number of valence electrons increases going left to right across a period. The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). The general trend is that radii increase down a group and decrease across a period. Valence electrons on the periodic table Bohr models for the first three periods of the periodic table are shown below. (b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. The highest occupied electron shell is 2, so the period number is 2. 
The periodic table and trends in valence electrons can be used to determine the number of bonds an atom is most likely to form. Atoms share electrons and form covalent bonds to satisfy the octet rule. Thus, as size (atomic radius) increases, the ionization energy should decrease.\): (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. 4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. For larger atoms, the most loosely bound electron is located farthest from the nucleus and so is easier to remove. This will be how many valence electrons there are. Counting along the row/period, count the number of boxes to your element. Locate the element on the periodic table. Energy is always required to remove electrons from atoms or ions, so ionization processes are endothermic and IE values are always positive. The valence electrons are found based on where the element is on the periodic table. The energy required to remove the third electron is the third ionization energy, and so on. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
